The Science
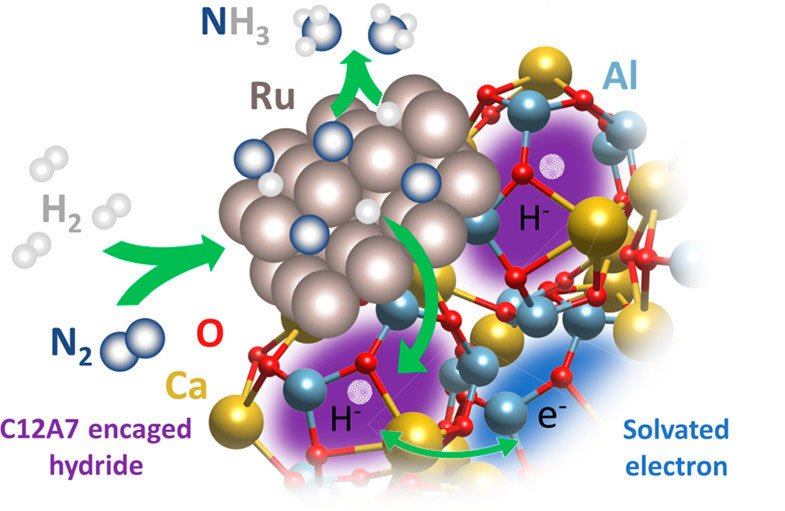
Neutron scattering unveiled new insights into the performance of a novel metal catalyst used to convert nitrogen into ammonia. Scientists use neutron scattering to learn about materials based on how atoms of those materials disperse neutrons. The key discovery is that the hydrogen atoms on the surface of the material play the most significant role in the ammonia synthesis. Previously, researchers believed the key atoms were those caged inside the catalyst.
The Impact
Ammonia is one of the most essential and widely produced chemicals in the world. It is an ingredient in fertilizers and used to produce many types of plastics and fabrics. Industries synthesize ammonia using catalysts, materials that speed up chemical reactions. This research involved a catalyst material made of ruthenium, calcium, and aluminum. The novel material catalyzes ammonia synthesis with significantly less energy (heat and pressure) than the traditional iron-based catalysts. This novel material could improve the efficiency of goods that require ammonia. This could lead to lower prices and higher quality products.
Summary
Catalysts enable desired chemical reactions, a process called catalysis. Catalysts come in many shapes, sizes, and materials. The most familiar example is the catalytic converter used in automobiles to convert carbon monoxide emissions into less harmful carbon dioxide. Industry uses catalysts for myriad applications including fuels, plastics, and medicines. Because ammonia is used in the fertilizers to grow crops, ammonia synthesis is an extremely important chemical process. Current technologies use mostly the iron-based catalysts in ammonia synthesis and the processes are energy intensive. Novel electride-based catalysts offer a greener approach, requiring significantly less heat and pressure (and thus less energy overall) for the reaction to proceed. Electride compounds contain excess electrons that play a chemical role in ammonia synthesis by weakening the nitrogen bonds. However, the precise mechanism by which electride catalysts work remains less understood.
Neutrons’ sensitivity to hydrogen and their ability to peer deeply inside materials make them ideal for studying catalytic reactions. Using the VISION instrument at Oak Ridge National Laboratory’s Spallation Neutron Source, along with computer modeling, researchers confirmed the catalyst’s primary reaction mechanism occurs due to the surface hydrogens of the catalyst, not those encaged inside the metal. This discovery paves the way for further studies to optimize the catalyst’s potential for higher performance.
Funding
This research was supported by the Laboratory Directed Research Development program at Oak Ridge National Laboratory (ORNL). The neutron studies were conducted at the Spallation Neutron Source, a Department of Energy (DOE) Office of Science user facility. Part of the catalyst synthesis and SEM imaging were conducted at the Center for Nanophase Materials Sciences and calculations used the National Energy Research Scientific Computing Center; both are DOE Office of Science user facilities. Also used in the research was the ORNL Compute and Data Environment for Science supported by the DOE Office of Science.