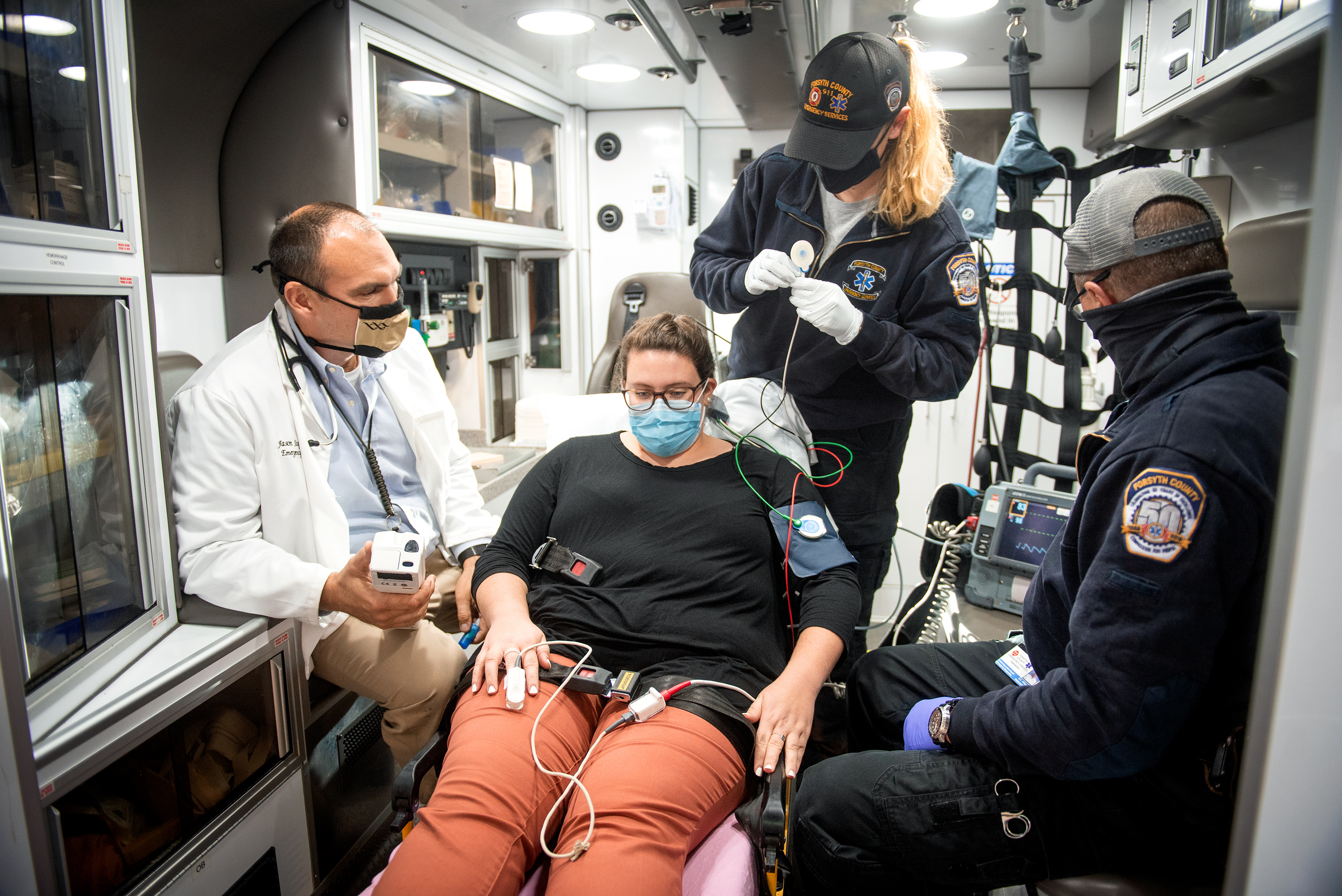
WINSTON-SALEM, N.C. – Oct. 7, 2020 – Is this person with chest pain having a heart attack? That’s a question EMTs frequently confront when responding to 911 calls.
A study conducted at Wake Forest Baptist Health shows that on-scene use of a new protocol and advanced diagnostic equipment can help paramedics better identify patients at high risk for adverse cardiac events. This approach could help paramedics determine the hospital best equipped to treat those people.
The study is published in the Oct. 7 edition of the journal PLOS One.
“While only 7% of people who make 911 calls due to chest pain are having a heart attack, paramedics must be able to make the correct decisions using objective measurements to identify another 20% that need specific cardiac care,” said principal investigator Jason Stopyra, M.D., associate professor of emergency medicine at Wake Forest Baptist. “High-risk patients are often transported to facilities that don’t have interventional cardiology capabilities, and later have to be transferred to another hospital for urgent procedures.”
Currently, pre-hospital assessment of patients with chest pain focuses on looking for signs of a heart attack with a portable electrocardiogram (EKG). For the 93% of people who aren’t having a discernible heart attack, paramedics rely on their experience and patient preference to decide what hospital to go to rather than on an objective risk assessment of the level of cardiac care needed, Stopyra said.
In the prospective study that took place from December 2016 to January 2018, the Wake Forest Baptist team worked with EMS agencies in three rural and urban counties in North Carolina to evaluate the use of a pre-hospital modified HEART Pathway (PHMP) to identify both high- and low-risk patients who called 911 with chest pains.
The HEART Pathway, which was developed by senior author Simon Mahler, M.D., professor of emergency medicine at Wake Forest Baptist, has been used in emergency departments for 8 years, Stopyra said.
The research team trained more than 150 paramedics to use a hand-held i-STAT, a modified version of a hospital device that measures patients’ blood for levels of troponin, a cardiac enzyme in the blood that can help detect heart injury. In addition, paramedics were trained to ask patients questions about their chest pain, age, risk factors and medical history to determine what’s known as a HEART risk score.
The patients in the study were stratified into three groups: high risk based on elevated troponin, low risk based on a HEART score of less than 4 with negative troponin, or moderate risk based on a HEART score of 4 or higher with negative troponin. Major adverse cardiac events (MACE) were determined by reviewing records after 30 days. The sensitivity and negative predictive values for MACE at 30 days also were calculated.
The study showed that a prospective application of the prehospital modified HEART Pathway, including measurement of troponin levels completed by paramedics during ambulance transport, achieved high sensitivity, specificity and negative predictive value for 30-day MACE. A high-risk assessment resulted in a specificity of 96% and positive predictive value of 61%. A low-risk assessment was associated with a negative predictive value of 94% and a specificity of 90%.
“This teamwork between emergency medicine researchers and our local EMS agencies is so valuable,” Stopyra said. “Our study showed that paramedics have the professional clinical ability to effectively assess patients’ cardiac risk using this protocol before the patient ever gets to the hospital. We hope this approach will allow patients to receive faster, more focused care.”
The study was limited in that it was observational, and no treatment decisions were made based on the PMHP made by the paramedics.
The i-STAT device used in this study was manufactured by Abbott Point of Care in Princeton, N.J., which also funded the study.