The benefit of osimertinib treatment demonstrated earlier this year in the ADAURA trial was so substantial that the independent data monitoring committee recommended early unblinding of the multinational randomized controlled phase III trial. The latest findings are to be presented September 19 at 12:30 p.m., during the virtual science program at the annual meeting of the European Society for Medical Oncology (ESMO). The results are also to be published online in the New England Journal of Medicine at the same time.
“These updated results from the ADAURA trial once again demonstrated a statistically significant and clinically meaningful improvement in disease-free survival in the adjuvant treatment of patients with early-stage EGFR mutations for NSCLC,” said Roy S. Herbst, MD, PhD, chief of Medical Oncology at YCC and Smilow Cancer Hospital and senior author of the study. “It’s so critical to provide patients with this type of lung cancer a new treatment option.”
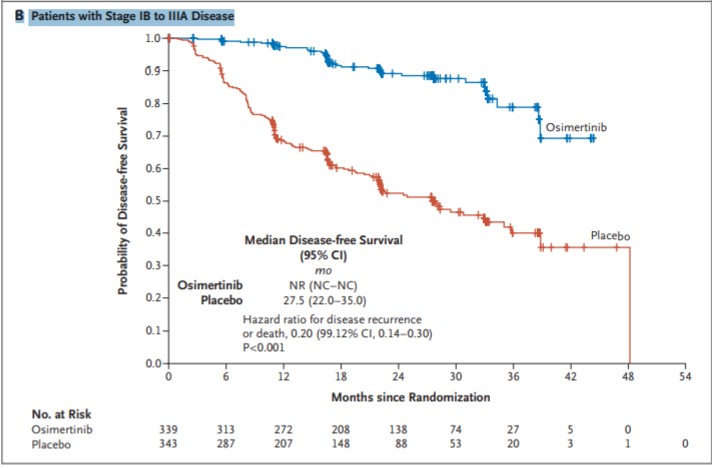
ADAURA is a phase 3, randomized trial that looked at adjuvant therapy with osimertinib, a third-generation EGFR-TKI, versus placebo, in treating patients with Stage IB through IIIA EGFR-mutated non-small cell lung cancer. The trial results showed that osimertinib offered a two-year, 89% DFS for patients with resected lung cancer (stage IB/II/IIIA) compared to a DFS rate of 52% in patients randomized to treatment with placebo, with manageable side effects. Disease-free survival measures the time from randomization to first sign of cancer recurrence or death. In this trial, patients treated with osimertinib had a 79% reduction in the risk of their cancer returning or death. The study will continue to follow patients for overall survival outcomes.
“In the past, we haven’t had much success fighting recurrence in the liver, lung and brain of NSCLC in patients with EGFR mutations,” said Herbst. “These study results will hopefully be practice-changing and have a huge impact on patient care.”
This study was funded by AstraZeneca.
About Yale Cancer Center and Smilow Cancer Hospital Yale Cancer Center (YCC) is one of only 51 National Cancer Institute (NCI-designated comprehensive cancer) centers in the nation and the only such center in Connecticut. Cancer treatment for patients is available at Smilow Cancer Hospital through 13 multidisciplinary teams and at 15 Smilow Cancer Hospital Care Centers in Connecticut and Rhode Island. Smilow Cancer Hospital is accredited by the Commission on Cancer, a Quality program of the American College of Surgeons. Comprehensive cancer centers play a vital role in the advancement of the NCI’s goal of reducing morbidity and mortality from cancer through scientific research, cancer prevention, and innovative cancer treatment.
# # #