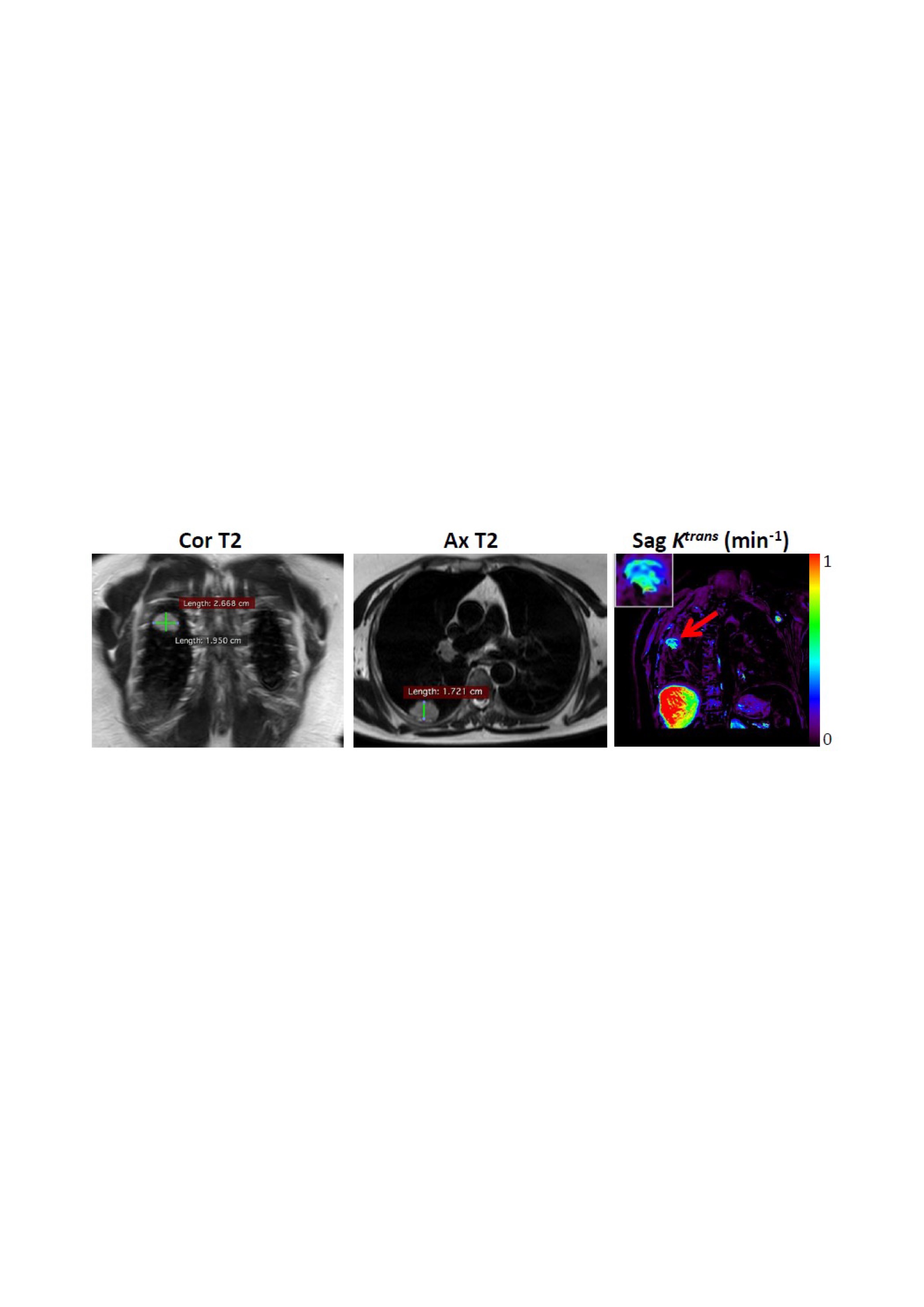
When the team of researchers and clinicians measured how much itraconazole ended up in the bloodstreams and tumors of 13 patients treated for lung cancer, they found a sixfold variation in drug levels in tumor samples. Moreover, the levels in the patients’ bodies correlated with how effectively the drug shrank their tumors.
“What this means going forward is that, in future studies of itraconazole for the treatment of cancer, it may be important to check each patient’s drug level and tailor the dose,” says David Gerber, M.D., a professor of internal medicine and population and data sciences at UTSW and first author of the new paper, published online in the journal Clinical Cancer Research. “In this context, there’s no one-size-fits-all dose,” notes Gerber, also Associate Director of Clinical Research in the Harold C. Simmons Comprehensive Cancer Center.
Itraconazole, sold as Sporonox, Sporaz, or Orungal, has been used for more than 25 years to treat fungal infections. Ten years ago, James Kim, M.D., Ph.D., an associate professor of internal medicine at UTSW and senior author of the study, was part of a team to discover that the antifungal drug also shuts down pathways used by cancer cells to grow. Further research has shown that itraconazole may help treat lung, prostate, skin, and other cancers by both blocking cellular growth pathways and stopping the formation of new blood vessels initiated by cancers.
“There was growing evidence that itraconazole conveyed a survival advantage to patients,” says Kim. “But, in this new work, we wanted to take a step back and look more at the biology and pharmacology of what was going on with this drug in cancer patients.”
For infections, itraconazole is typically given in two 100 milligram doses per day. Other cancer trials have used doses of the drug ranging from 200 to 600 milligrams per day. Gerber, Kim, and other colleagues studied the effect of a steady dose of itraconazole – 300 milligrams twice a day with food – in 13 patients with non-small cell lung cancer who were already scheduled for surgery to remove their tumors. After each surgery, the researchers analyzed resected tumor samples to determine how much itraconazole had accumulated in the cancer cells.
Levels of itraconazole detected within patients’ tumors ranged from 1,244 ng/g to 7,094 ng/g. This nearly sixfold variation could not be fully explained by factors known to affect drug dosing, including body mass and kidney or liver function. Over the 14-day treatment period, change in tumor size ranged from a 26 percent decrease to 13 percent growth. Patients with the highest blood and tumor levels of itraconazole also had the largest decreases in their tumor volumes.
Further tissue analysis and imaging studies revealed corresponding changes in the growth of tumor blood vessels and blood flow; the patients with higher levels of itraconazole also had greater reductions in both of these parameters.
“This study highlights the need, when repurposing drugs, to look closely at the dosing,” says William Trey Putnam, Ph.D., director of the Clinical Pharmacology Center at Texas Tech University Health Sciences Center, who collaborated with Gerber and Kim on the research. “In different diseases, the dosing can end up needing to be quite different.”
The current study was not designed to look for side effects, but the researchers say the dose being used was within the range previously determined to be safe.
Because it’s been used as an antifungal drug for decades, itraconazole is significantly cheaper than most other cancer drugs that have similar molecular effects on tumor growth and tumor blood vessels. The researchers say future studies will examine the use of itraconazole in combination with other cancer drugs to reveal why the drug is processed so differently by different patients.
“We had a small number of patients enrolled in this trial, but we were able to optimize the use of specimens and clinical data to get statistically significant results,” says Farjana Fattah, Ph.D., a UTSW assistant professor with the Harold C. Simmons Comprehensive Cancer Center who helped lead the research. “Larger studies might be able to draw even more conclusions.”
Other UTSW researchers who contributed to this study were Kemp Kernstine, Rolf Brekken, Ivan Pedrosa, Rachael Skelton, Jessica Saltarski, Robert Lenkinski, Chul Ahn, Chyndhri Padmanabhan, Vaidehi Chemburkar, Sahba Kasiri, Qing Yuan, Quyen Do, Yin Xi, Scott Reznik, Lorraine Pelosof, Brandon Faubert, Ralph DeBerardinis, and James Kim. Collaborators were Richard Leff, Rajareddy Kallem, and Indhumathy Subramaniyan of Texas Tech University.
This research was supported by funds from the Department of Defense Lung Cancer Research Program (W81XWH-14-1-0540, W81XWH-14-1-0338, the National Cancer Institute (K24CA201543-01, 1R01CA196851, R35CA22044901), the American Cancer Society (RSG-16-090-01-TBG), the Cancer Prevention and Research Institute of Texas Core Facilities Support Awards (RP170003), the Canadian Institutes of Health Research (MFE 140911), the Cancer Prevention and Research Institute of Texas (RP160089), and the National Heart, Lung, and Blood Institute (5T32HL098040).
About UT Southwestern Medical Center
UT Southwestern, one of the premier academic medical centers in the nation, integrates pioneering biomedical research with exceptional clinical care and education. The institution’s faculty has received six Nobel Prizes, and includes 23 members of the National Academy of Sciences, 16 members of the National Academy of Medicine, and 13 Howard Hughes Medical Institute Investigators. The full-time faculty of more than 2,500 is responsible for groundbreaking medical advances and is committed to translating science-driven research quickly to new clinical treatments. UT Southwestern physicians provide care in about 80 specialties to more than 105,000 hospitalized patients, nearly 370,000 emergency room cases, and oversee approximately 3 million outpatient visits a year.