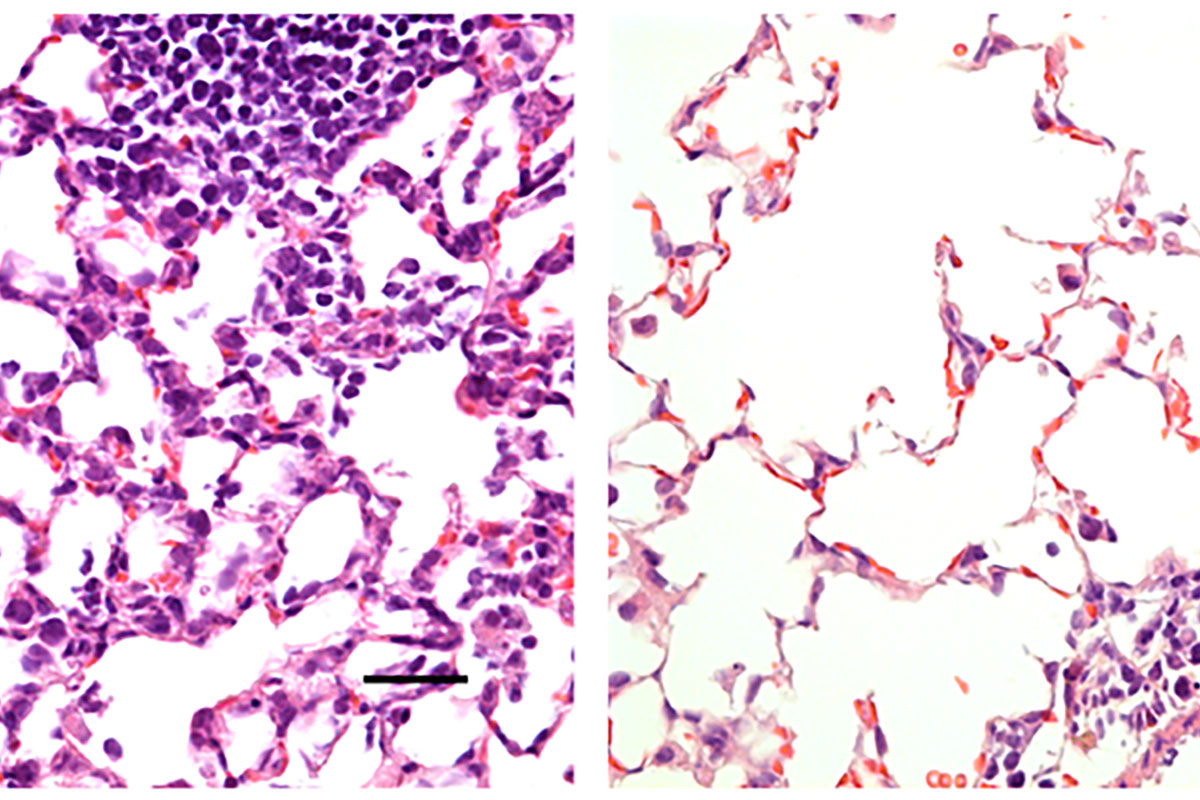
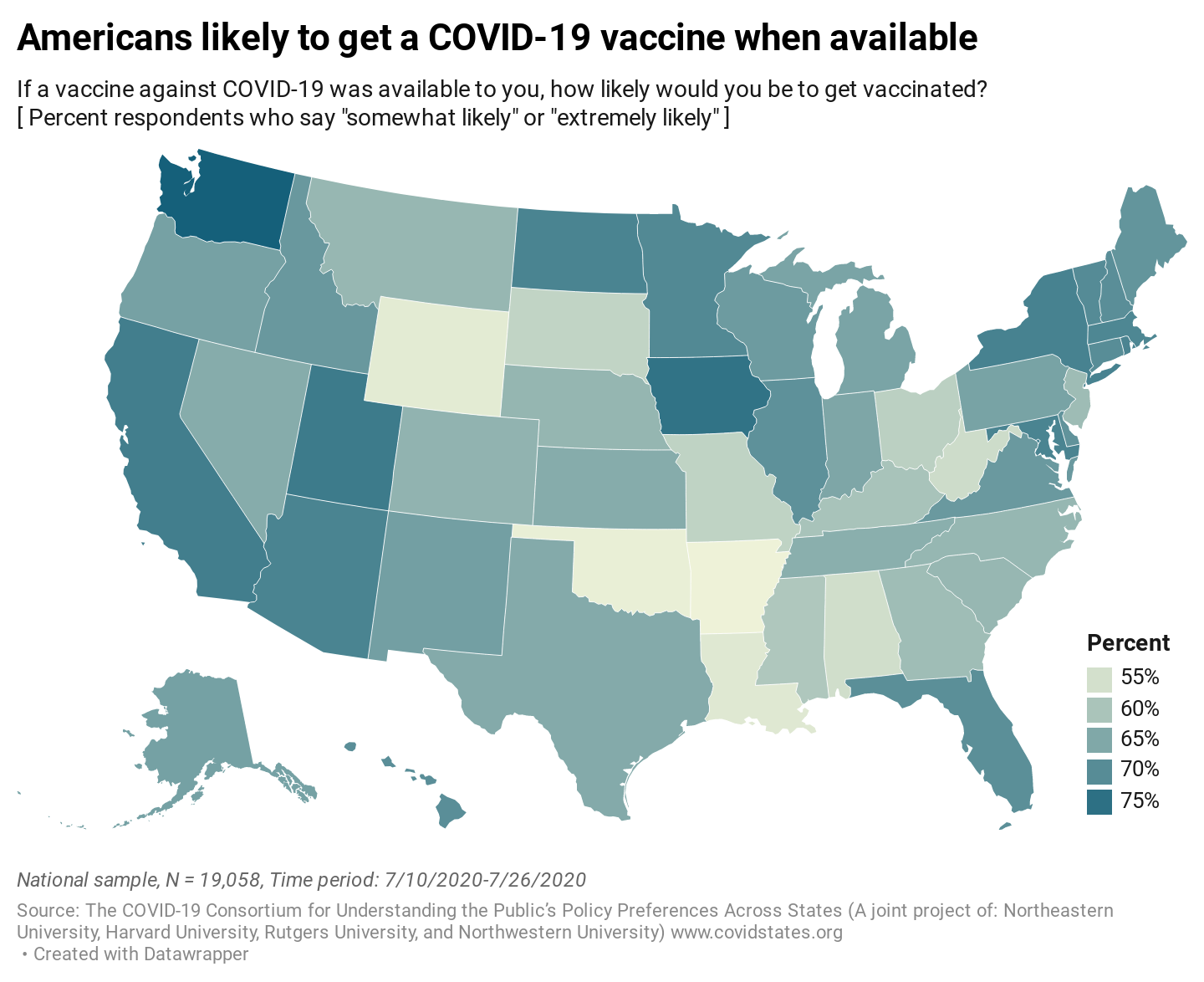
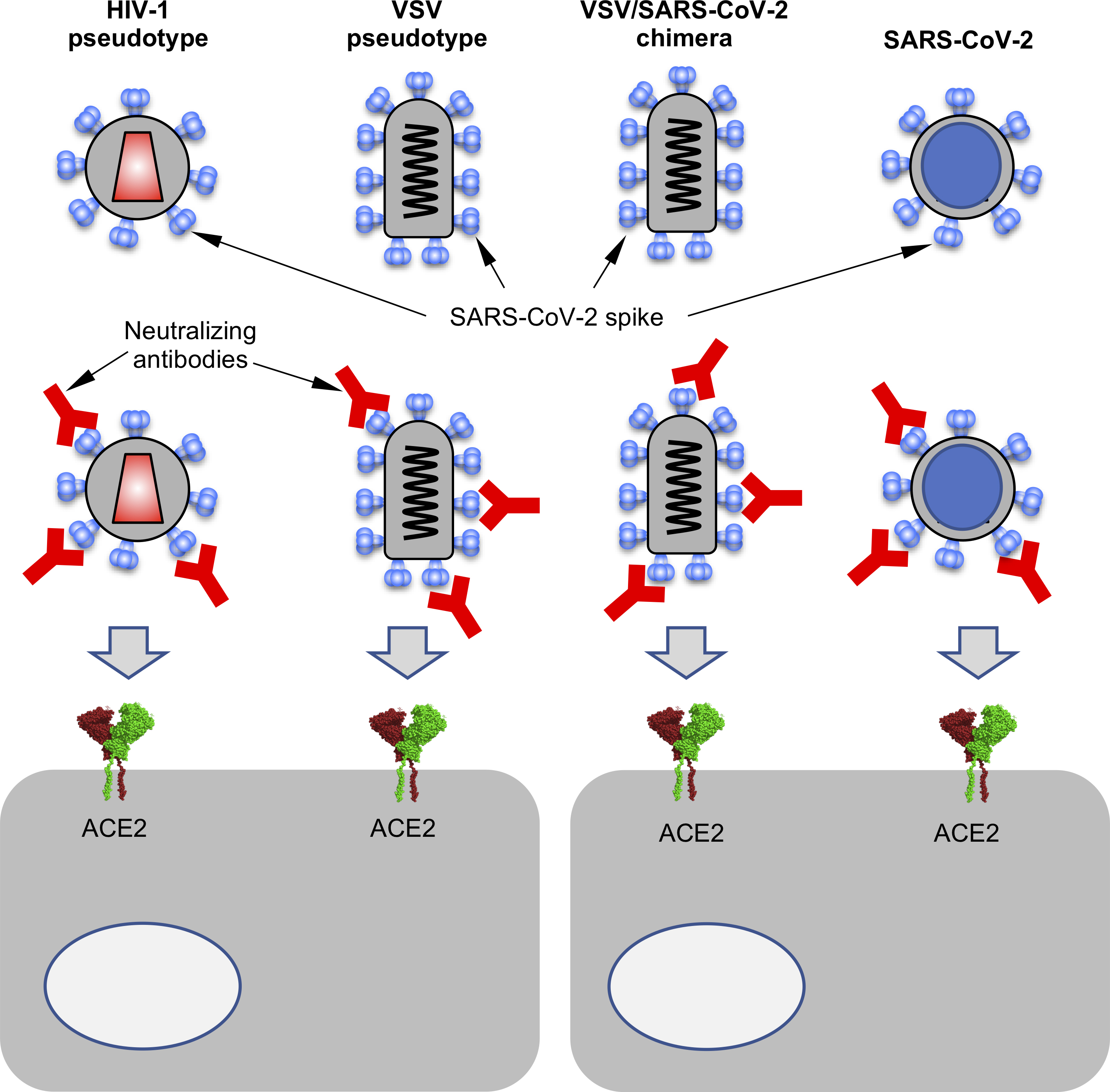
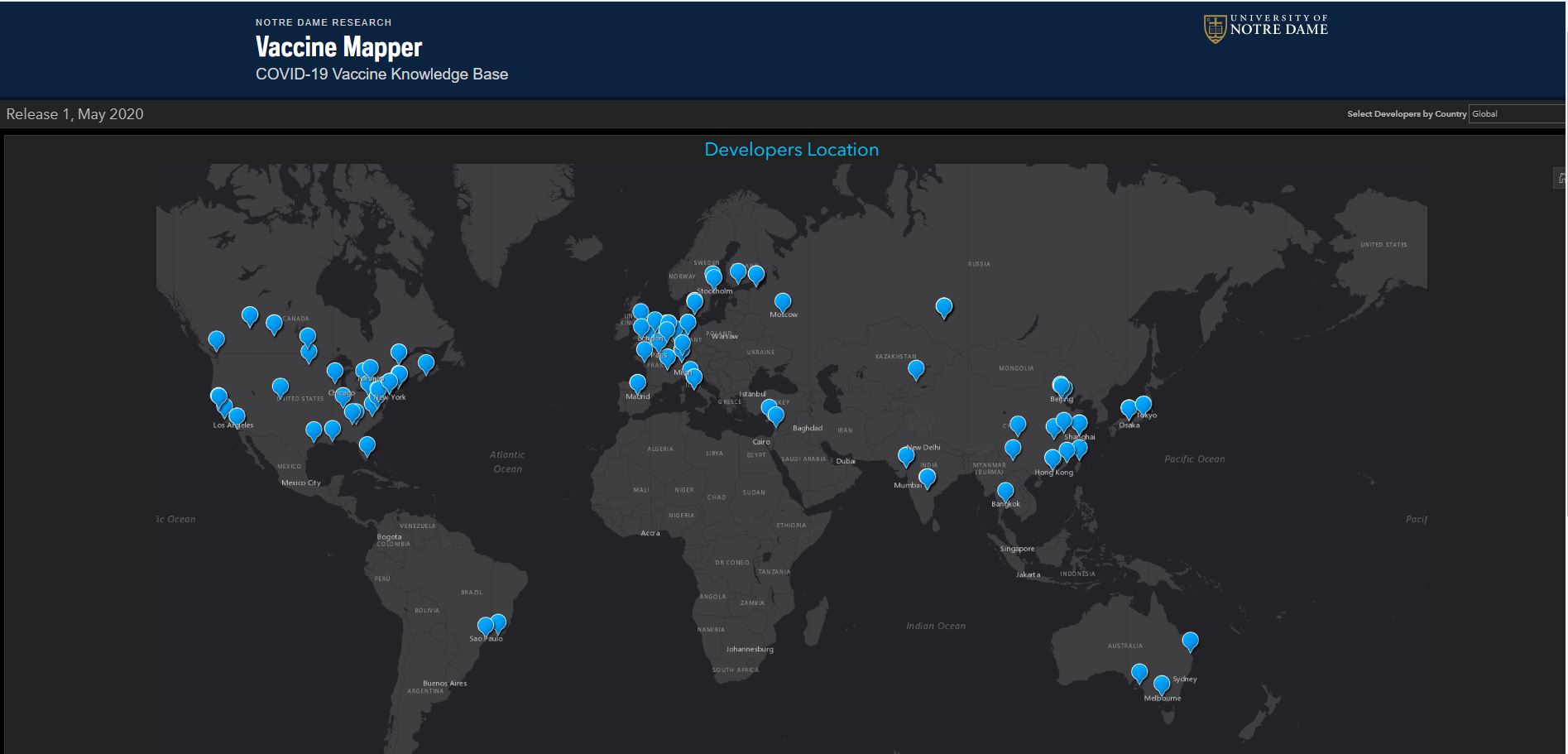
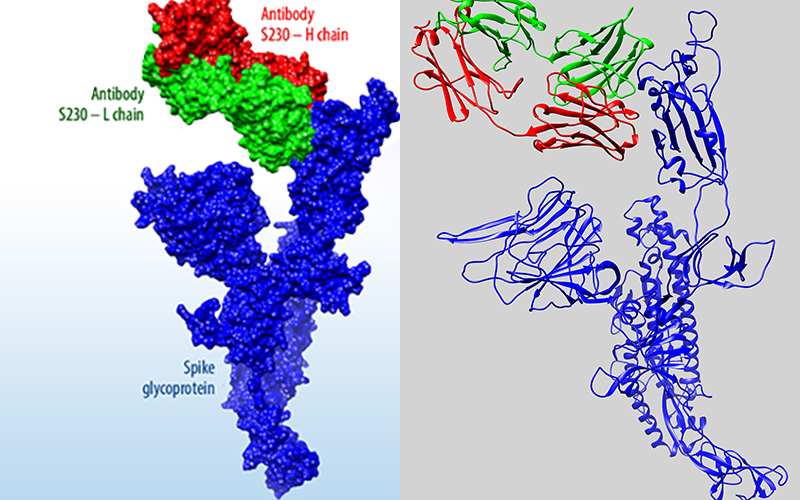
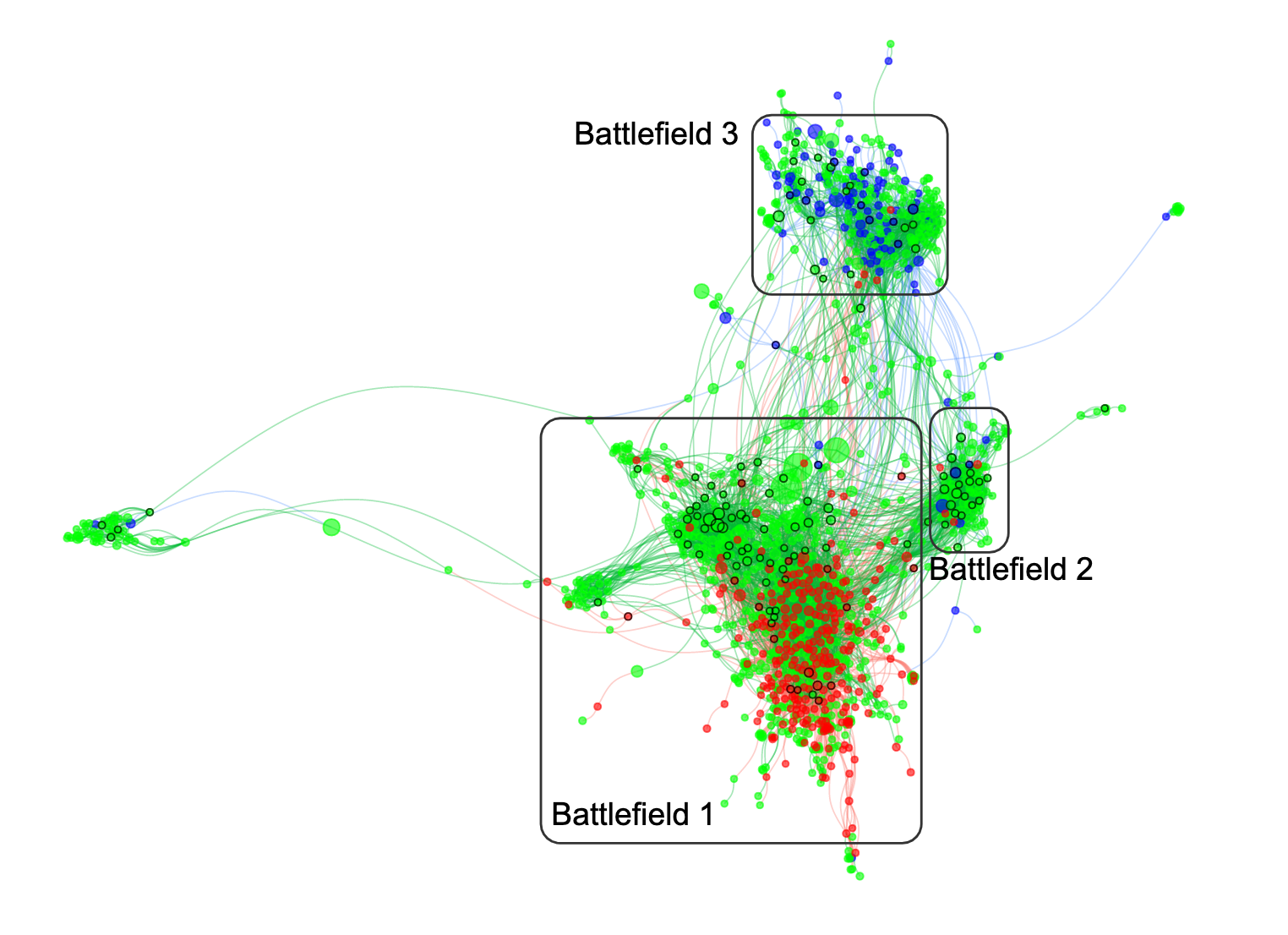
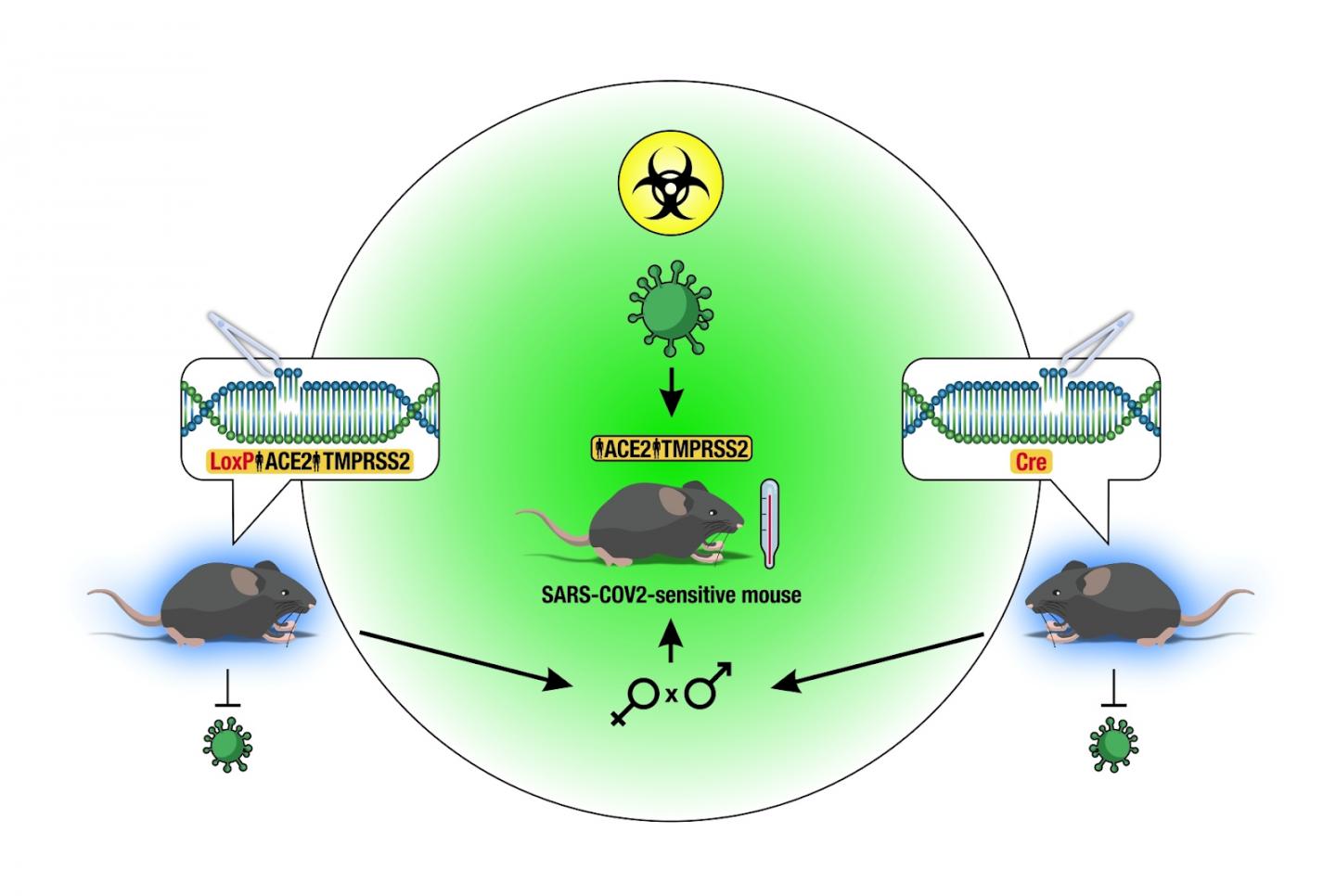
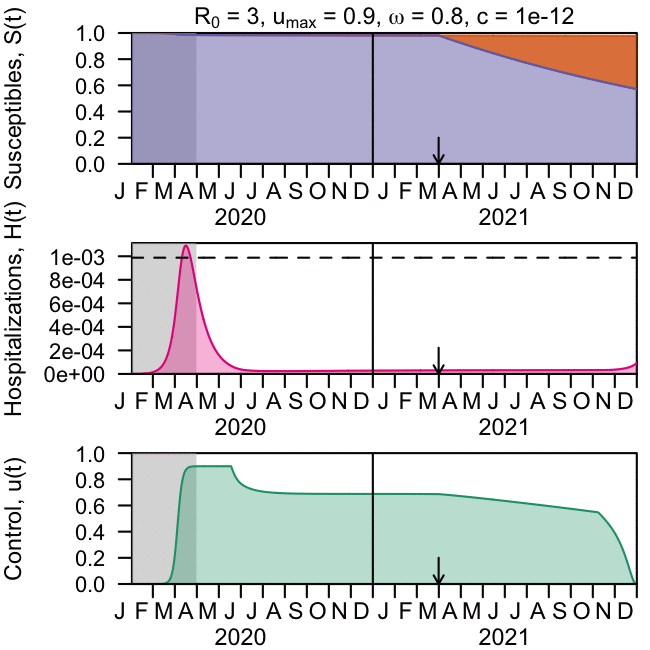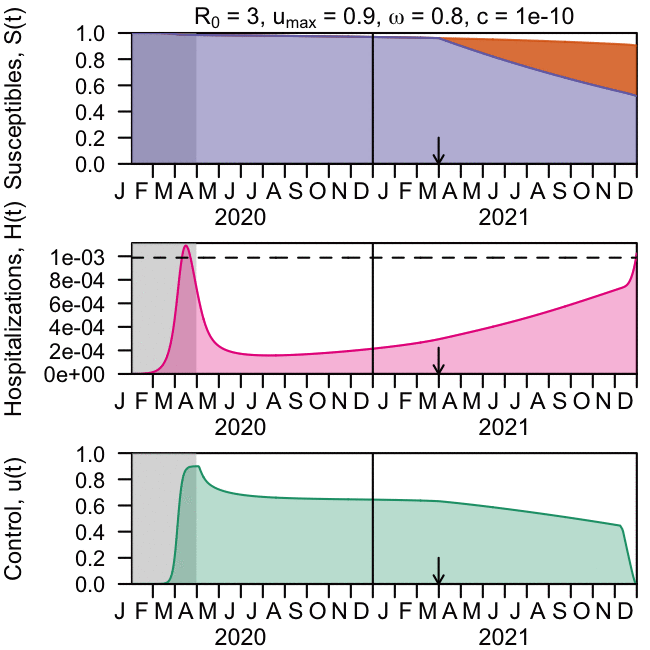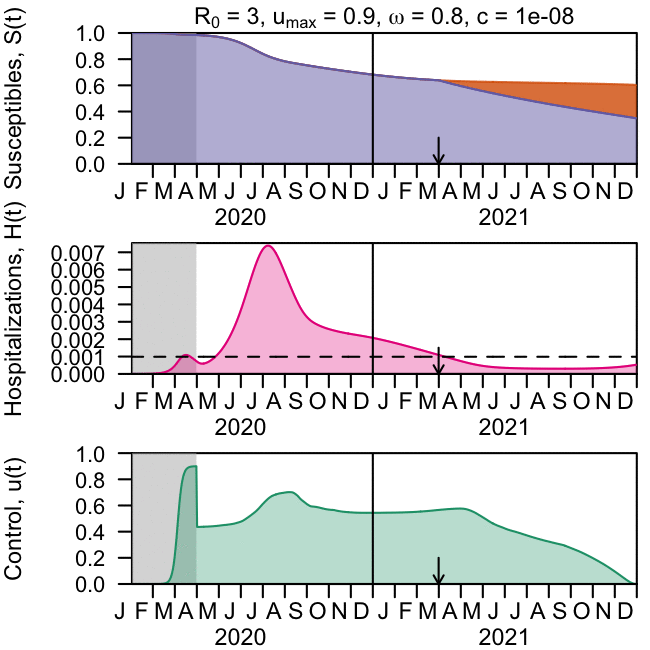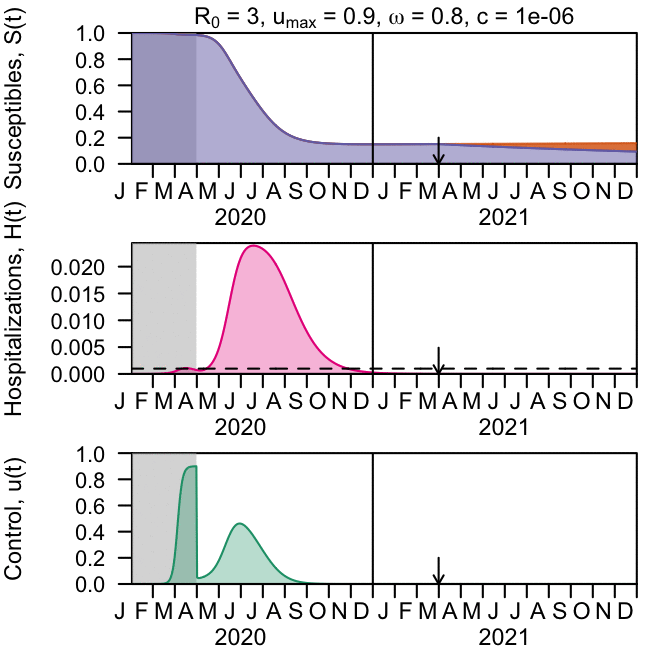
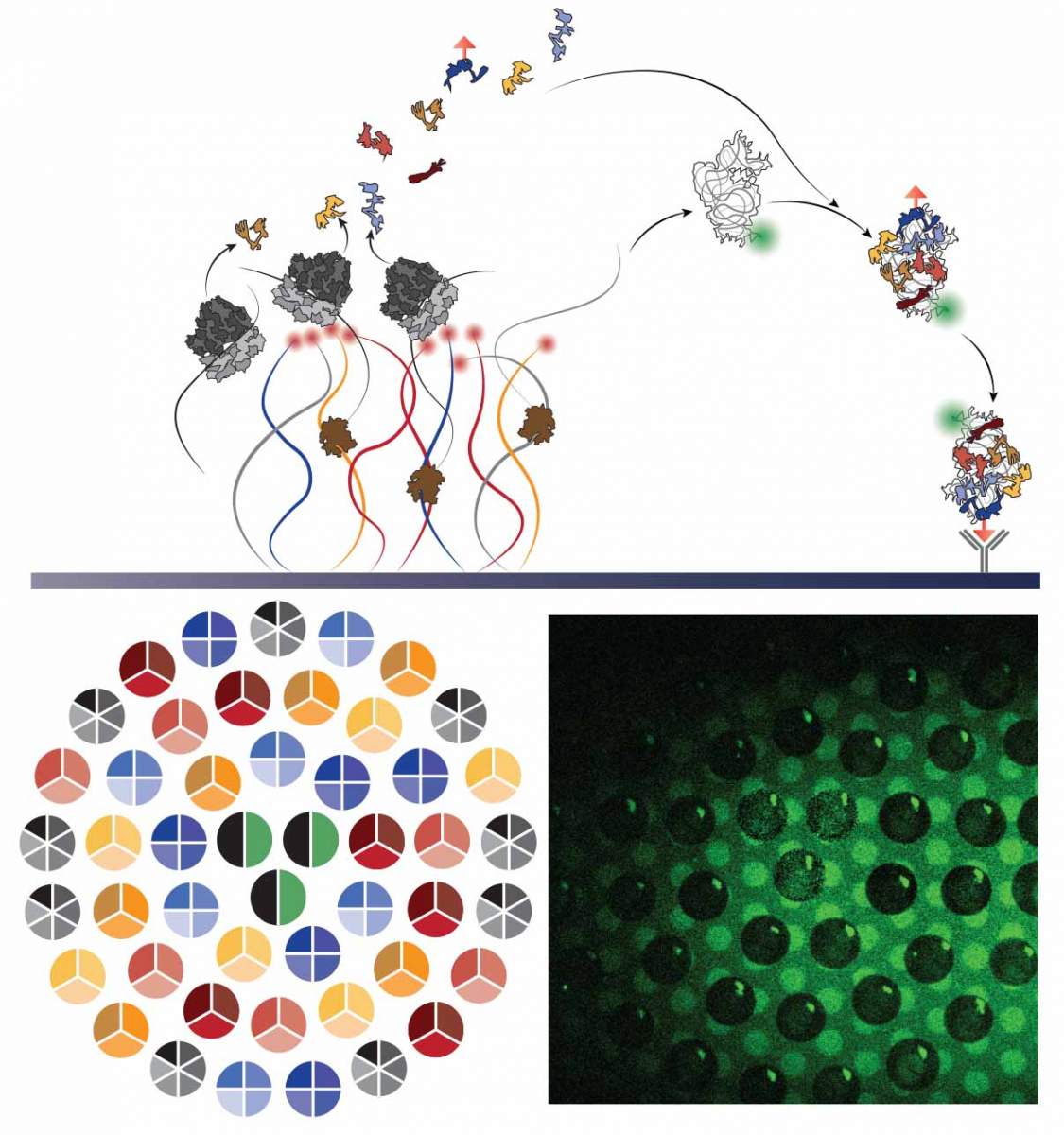
As the global response to the SARS-COV-2 virus that causes COVID-19 approaches 200 days, Baylor Scott & White Research Institute, the research and development arm of Baylor Scott & White Health, is accelerating its pace of bringing clinical trials online.
Baylor Scott & White Research Institute continues to mobilize staff and resources, including components needed to integrate critical patient-safety measures at every participating site within the Baylor Scott & White system for industry sponsored drug trials, investigator-initiated drug trials and research studies, and observational and data studies designed to help increase knowledge around case trends, viral epidemiology, and care best practices.