Monitoring levels of DNA shed by tumors and circulating in the bloodstream could help doctors accurately assess how gastroesophageal cancers are responding to treatment, and potentially predict future prognosis, suggests a new study led by researchers at the Johns Hopkins Kimmel Cancer Center and its Bloomberg~Kimmel Institute for Cancer Immunotherapy.
Tag: liquid biopsy

Pediatric Cancer Expert Explains New Options for Children With Sarcomas
Leo Mascarenhas, MD, MS, has an important message for parents whose children have been diagnosed with sarcoma, a type of cancer that develops in the bones or soft tissues.
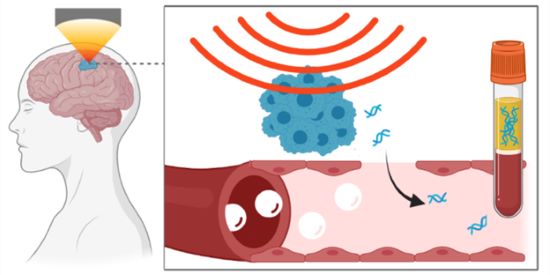
Bypassing the blood-brain barrier to improve brain tumor diagnosis
A collaborative team of NIH-funded researchers is developing a way to obtain DNA shed from brain tumors using focused ultrasound. Their first-in-human study could be an important step towards improving the way brain tumors are diagnosed.
Dana-Farber and Gustave Roussy to hold third Transatlantic Exchange: Annual scientific conference dedicated to advancing Oncology research and practice
Dana-Farber Cancer Institute (Boston, MA, USA) and Gustave Roussy (Grand Paris, Villejuif, France) have announced that the third Transatlantic Exchange in Oncology Conference will address: Liquid Biopsy as an Emerging Approach in Precision Cancer Medicine.The meeting, supported by L’Institut Servier, will be held in-person (witha virtual attendance option) on April 12, 2024, at the Revere Hotel Boston Common and livestreamed virtually on Medscape’s platform.
Immune Cell Receptor Provides Promising Immunotherapy Target
Drugs that target a receptor on immune cells called activin receptor 1C may combat tumor-induced immune suppression and help patients’ immune systems fight back against cancer, according to a study by investigators at the Johns Hopkins Kimmel Cancer Center and its Bloomberg~Kimmel Institute for Cancer Immunotherapy.
Association for Molecular Pathology Publishes Best Practice Recommendations for Liquid Biopsy Assay Validations
The Association for Molecular Pathology has published best practice recommendations for validating and reporting clinical circulating tumor DNA (ctDNA) or liquid biopsy assays and their related scientific publications.
Capturing Immunotherapy Response in a Blood Drop
Liquid biopsies are blood tests that can serially measure circulating tumor DNA (cell-free DNA that is shed into the bloodstream by dying cancer cells). When used in patients with advanced non-small cell lung cancer undergoing immunotherapy, they may identify patients who could benefit from treatment with additional drugs, according to a phase 2 clinical trial in the U.S. and Canada. The trial is led by investigators at the Johns Hopkins Kimmel Cancer Center and its Bloomberg~Kimmel Institute for Cancer Immunotherapy, BC Cancer and the Canadian Cancer trials Group (CCTG).
Cancer: In Italy every year over 8000 patients candidate for liquid biopsy
SHRO Founder Antonio Giordano contributes to a new publication on the use of liquid biopsy to assess the treatment response and find mutations that confer resistance to cancer therapies.
Researchers test cost-effective approach to early-cancer detection from cell-free DNA in blood samples
Researchers at UCLA’s Jonsson Comprehensive Cancer Center and collaborating organizations report successful results from an experimental cancer-detection system that appears to have overcome some of the challenges of the “liquid biopsy” in a novel, cost-effective way.
Children’s Hospital Los Angeles Identifies Molecular Markers to Increase Precision in Treatment of Retinoblastoma
Retinoblastoma is a cancer of the eye that occurs in infants and toddlers and can lead to loss of vision, loss of one or both eyes, and even death. Unlike most tumors, these cannot be biopsied because of the risk of spreading cancer to the rest of the body. In 2017, Jesse Berry, MD, surgeon and ocular oncologist at Children’s Hospital Los Angeles, discovered that fluid removed from the eye during treatment of retinoblastoma contained tumor DNA and functioned as a liquid biopsy—providing information about the tumor and opening the door to earlier diagnosis and treatment.
DNA Shed From Colon Cancers Into Bloodstream Successfully Guides Chemotherapy After Surgery
A multi-institutional, international study, led by researchers at the Johns Hopkins Kimmel Cancer Center and WEHI in Melbourne, Australia, found that testing for ctDNA after surgery and directing chemotherapy to ctDNA-positive patients reduced the use of chemotherapy overall without compromising recurrence-free survival.
Biomarker in Liquid Biopsy for Lung Cancer Appears More Accurate in Predicting Immunotherapy Response Than Tumor Biopsy
Mount Sinai researchers have validated for the first time that a simple blood test called a liquid biopsy could be a better predictor of whether cancer immunotherapy will be successful for a patient with lung cancer than an invasive tumor biopsy procedure. Their study was published in the Journal of Experimental & Clinical Cancer Research in June.
Liquid biopsies may aid diagnosis, treatment of bladder, nerve tumors
Two studies led by Washington University School of Medicine in St. Louis describe the potential of liquid biopsies to identify and track tumor growth in two very different cancers: bladder cancer and peripheral nerve tumors.
NUS researchers develop world’s first blood test for real-time monitoring of cancer treatment success
A team of researchers from the NUS Department of Biomedical Engineering and Institute for Health Innovation & Technology has developed a novel blood test called ExoSCOPE that could tell doctors whether cancer treatment is working for a patient, within 24 hours after the treatment. This will enable doctors to customise the treatment plan to improve patients’ chances of recovery.

Liquid biopsy for colorectal cancer could guide therapy for tumors
A new study from Washington University School of Medicine in St. Louis demonstrates that a liquid biopsy examining blood or urine can help gauge the effectiveness of therapy for colorectal cancer that has just begun to spread beyond the original tumor. Such a biopsy can detect lingering disease and could serve as a guide for deciding whether a patient should undergo further treatments.

Cancer Research Institute and The Mark Foundation for Cancer Research Launch Collaboration to Evaluate Liquid Biopsy for More Accurate and Rapid Assessment of Lung Cancer Patient Response to Immunotherapy
The Cancer Research Institute (CRI) and The Mark Foundation for Cancer Research have launched a clinical trial that aims to demonstrate the utility of a novel, ultra-sensitive biomarker-directed blood test, or liquid biopsy, in assessing cancer patient responses to immunotherapy.

JNCCN Study Explores if Insurance is Keeping Pace with Trends in Targeted Cancer Therapy
New research from the University of California, San Francisco (USCF) and City of Hope in the July 2020 issue of JNCCN—Journal of the National Comprehensive Cancer Network examines coverage trends for circulating tumor DNA testing, also known as gene sequencing of ctDNA or “liquid biopsies.” The researchers found coverage rate rose from 0% to 38% in three years. The policies also increased in scope from 2017-2019, going from one cancer type to 12.
Biomarker test highly accurate in detecting early kidney cancer
A novel liquid biopsy method can detect kidney cancers with high accuracy, including small, localized tumors which are often curable but for which no early detection method exists, say scientists from Dana-Farber Cancer Institute.
The report in Nature Medicine suggests that if validated in larger trials and applied widely, the non-invasive test could find more early kidney cancers when they haven’t spread, thus reducing the mortality of the disease.
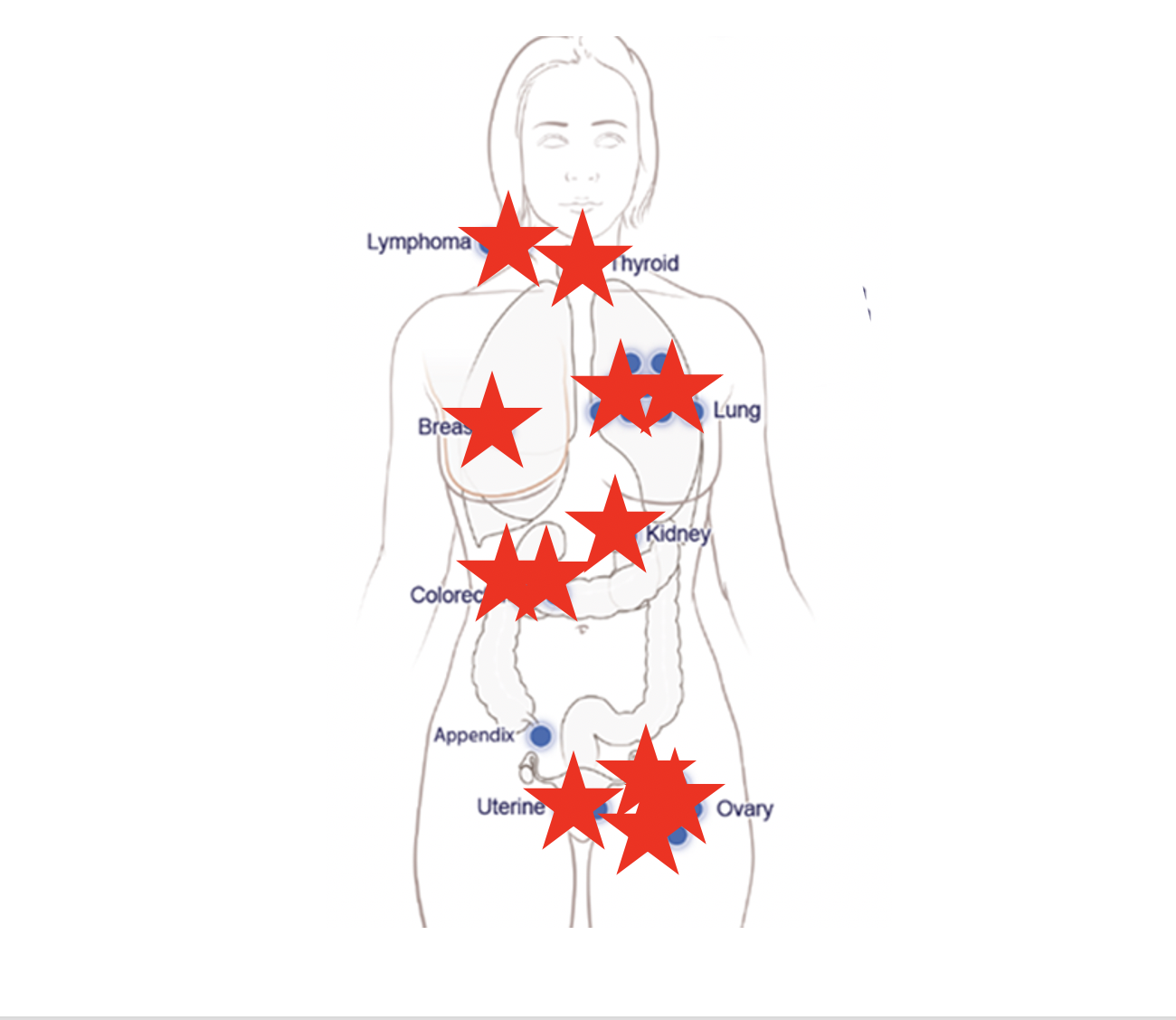
First study of multicancer blood test to screen for cancer guide intervention
Results from a first-of-its-kind study of a multicancer blood test in more than 9,900 women with no evidence or history of cancer showed the liquid biopsy test safely detected 26 undiagnosed cancers, enabling potentially curative treatment.
Microbial DNA in Patient Blood May be Tell-Tale Sign of Cancer
From a simple blood draw, microbial DNA may reveal who has cancer and which type, even at early stages
Intratumoral heterogeneity may be responsible for chemotherapy resistance in patients with small cell lung cancer
A study at The University of Texas MD Anderson Cancer Center found that chemotherapy results in increased heterogeneity within small cell lung cancer, leading to the evolution of multiple resistance mechanisms.

NUS researchers discover breakthrough in cancer diagnosis using big data analytics
A team of researchers from the National University of Singapore has developed a personalised assessment tool which can detect the incidence of cancer, predict patient survivability and determine patient suitability for immunotherapy cancer treatment.

Blood Test Can Predict Prognosis in Deadly Brain Cancer
A blood test that measures the amount of cell-free DNA (cfDNA) in the bloodstream – called a liquid biopsy – correlates with how patients will progress after they are diagnosed with glioblastoma (GBM), the deadliest and most common primary brain tumor in adults
MSK-ACCESS Receives New York State Approval for New Molecular Assay
Memorial Sloan Kettering Cancer Center (MSK) announced that the New York State Department of Health has issued an approval for a new molecular assay called Analysis of Circulating cfDNA to Evaluate Somatic Status (MSK-ACCESS). MSK-ACCESS was developed within the Marie-Josée and…