The U.S. Department of Energy (DOE) today announced up to $500 million in funding for basic research in support of DOE’s clean energy, economic, and national security goals.
Tag: Basic Energy Sciences
Department of Energy Announces $30 Million for Research to Accelerate Scientific Advances at User Facilities
Today, the U.S. Department of Energy (DOE) announced $30 million in funding for three projects to increase scientific productivity and discoveries across DOE light source, neutron source, and high-performance computing and networking facilities.
Department of Energy Announces $73 Million for Basic Research to Accelerate the Transition from Discovery to Commercialization
Today, the U.S. Department of Energy (DOE) announced $73 million in funding for eleven projects which focus on the goal of accelerating the transition from discovery to commercialization of new technologies that will form the basis of future industries.
Department of Energy Announces $112.4 Million for Research to Support National Biopreparedness and Response
Today, the U.S. Department of Energy (DOE) announced $112.4 million in funding for 10 research projects for the Biopreparedness Research Virtual Environment (BRaVE) initiative. These projects will support national biopreparedness and response capabilities that can be advanced with DOE’s distinctive capabilities.
DOE Awards $135 Million For Groundbreaking Research By 93 Early Career Scientists
The U.S. Department of Energy (DOE) today announced the selection of 93 early career scientists from across the country who will receive a combined $135 million in funding for research covering a wide range of topics, from artificial intelligence to astrophysics to fusion energy. The 2023 Early Career Research Program awardees represent 47 universities and 12 DOE National Laboratories across the country. These awards are a part of the DOE’s long-standing efforts to develop the next generation of STEM leaders to solidify America’s role as the driver of science and innovation around the world.
U.S. Department of Energy Announces $68 Million For Small Businesses Developing Technologies to Cut Emissions and Study Climate
The U.S. Department of Energy (DOE) today announced awards totaling more than $68 million that will go to 53 small businesses that are solving scientific problems. Projects include developing tools for climate research and advanced materials and technologies for clean energy conversion. Understanding the climate and the ability to convert and store energy are instrumental to meeting President Biden’s goal of a completely clean electrical grid by 2035 and net-zero greenhouse-gas emissions by 2050.
Add-on device makes home furnaces cleaner, safer and longer-lasting
Scientists at Oak Ridge National Laboratory have developed an affordable add-on acid gas reduction technology that removes 99.9% of acidic gases and other emissions to produce an ultraclean natural gas furnace. The AGR technology can also be added to other natural gas-driven equipment.
Department of Energy Announces $105 Million for Research to Support the Biopreparedness Research Virtual Environment (BRaVE) Initiative
Today, the U.S. Department of Energy (DOE) announced $105 million for research in biopreparedness. This funding, provided by the Office of Science, will support fundamental research to accelerate breakthroughs in support of the Biopreparedness Research Virtual Environment (BRaVE) initiative.
Department of Energy Announces $21 Million to Support Energy-Relevant Research in Underrepresented Regions
Today, the U.S. Department of Energy (DOE) announced $21 million in funding for 29 new projects through the Established Program to Stimulate Competitive Research (EPSCoR). By coupling innovative ideas from EPSCoR-eligible institutions with leading-edge capabilities at the DOE national laboratories, the grants are aimed to enhance the research of EPSCoR investigators while building expertise and capabilities that will enable the institutions to compete more successfully for other federal R&D funding. In this way, the DOE EPSCoR program advances the geographic diversity of researchers conducting competitive energy-related research.

Athena Safa Sefat: Then and Now
Athena Safa Sefat is a Senior Research Scientist and a former Wigner Fellow in the Materials Science & Technology Division of the Physical Sciences Directorate at Oak Ridge National Laboratory.

Making a Material World Better, Faster Now: Q&A With Materials Project Director Kristin Persson
Berkeley Lab’s Kristin Persson shares her thoughts on what inspired her to launch the Materials Project online database, the future of materials research and machine learning, and how she found her own way into a STEM career.
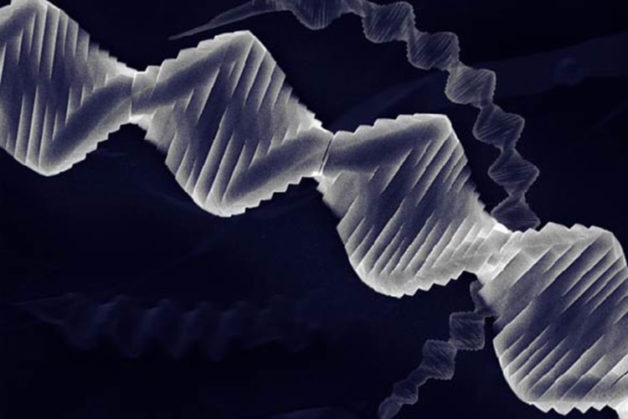
Crystal with a Twist: Researchers Grow Spiraling New Material
Scientists have created new inorganic crystals made of stacks of atomically thin sheets.

Machine Learning Accelerates Metamaterial Design
Researchers are increasingly using computer models to predict how light will interact with metamaterials. Scientists used machine learning techniques to analyze databases of information. The computer program predicted the ideal metamaterial design for absorbing low-energy light.

Improved Catalyst Branches Out and Out-Performs
Scientists can control their branch sizes and surfaces to make them more stable and more effective catalysts. By creating branched nanoparticles from the metal ruthenium, researchers developed a way to increase the speed of catalysis while maintaining the catalyst’s stability.

If You Want to Catch More Light, Twist It
Scientists created devices based on one Weyl semimetal, tantalum arsenide. They observed that this material was able to convert more light to electricity than any other material. The conversion was 10 times higher than previous measurements with other materials.
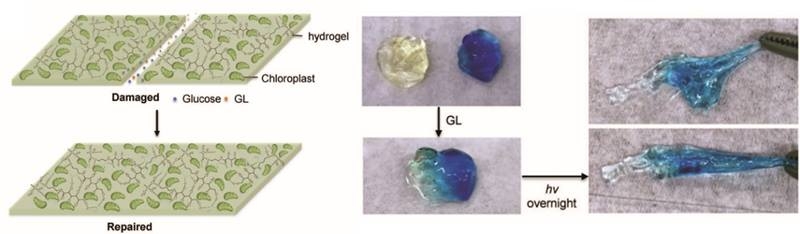
Growth and Repair from Carbon Dioxide in Air
Scientists have created a new class of material that uses sunlight to absorb and fix carbon dioxide from the atmosphere.
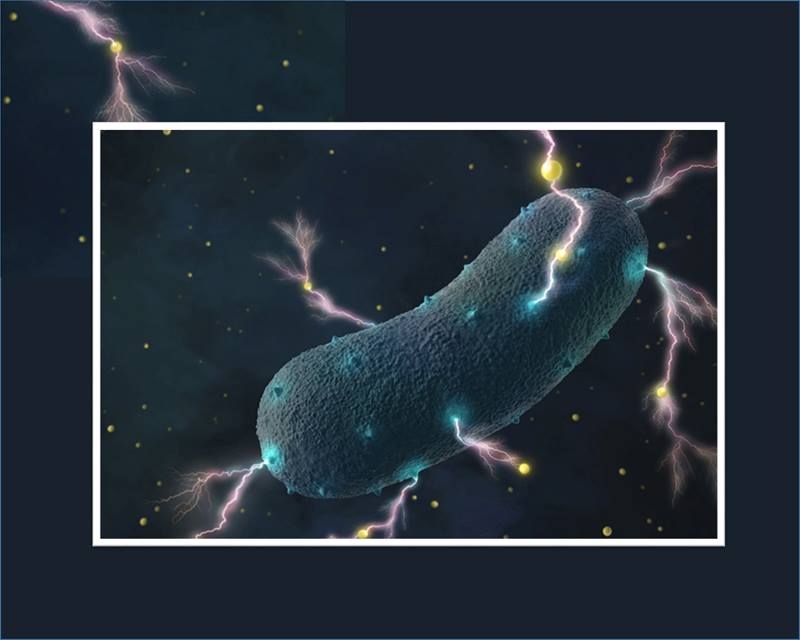
Gut Bacteria Found to Have a Shocking Secret
Scientists studied how the bacteria transport electrons across their cell wall. The bacteria use a method that’s different from other, known electricity-producing bacteria. They also found that hundreds of other bacterial species use this same process.

When Ions and Molecules Cluster
How an ion behaves when isolated within an analytical instrument can differ from how it behaves in the environment. Now, Xue-Bin Wang at Pacific Northwest National Laboratory devised a way to bring ions and molecules together in clusters to better discover their properties and predict their behavior.

Even Hard Materials Have Soft Spots
The Achilles Heel of “metallic glasses” is that while they are strong materials—even stronger than conventional steels—they are also very brittle. The initial failures tend to be localized and catastrophic. This is due to their random amorphous (versus ordered crystalline) atomic structure. Computer simulations revealed that the structure is not completely random, however, and that there are some regions in the structure that are relatively weak. Defects nucleate more easily in these regions, which can lead to failure. This understanding of the mechanical properties has led to a strategy for making the material stronger and less brittle.

2-D Atoms Do the Twist
In the study, scientists demonstrated, for the first time, an intrinsically rotating form of motion for the atoms in a crystal. The observations were on collective excitations of a single molecular layer of tungsten diselenide. Whether the rotation is clockwise or counter-clockwise depends on the wave’s propagation direction.
DOE Issues Small Business Research and Development Funding Opportunity Announcement
The Department of Energy (DOE) Small Business Innovation Research (SBIR) and Small Business Technology Transfer (STTR) programs issued its first Funding Opportunity Announcement (FOA) for Fiscal Year 2020.
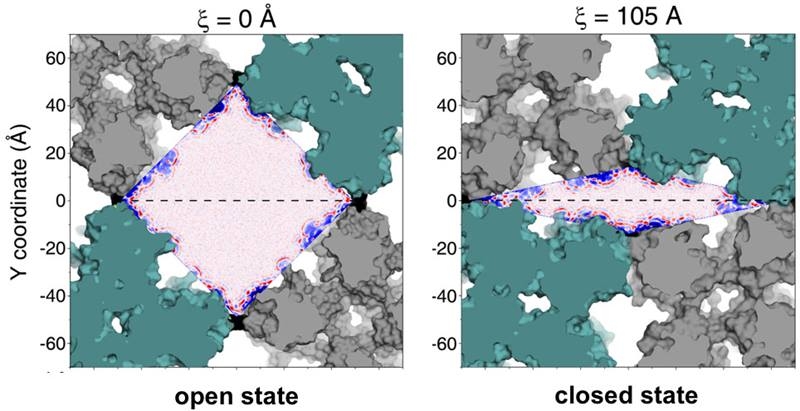
Cultivating the Assembly Landscape
For the first time, a team determined and predictably manipulated the energy landscape of a material assembled from proteins. Designing materials that easily and reliably morph on command could benefit water filtration, sensing applications, and adaptive devices.